LIDDELOW LAB
institute for translational neuroscience
@ NYU GROSSMAN SCHOOL OF MEDICINE
Astrocytes are among the most abundant cells in the mammalian central nervous system (CNS). They are integral to brain and spinal-cord physiology and perform many functions important for normal neuronal development, synapse formation, and proper propagation of action potentials. We still know very little, however, about how these functions change in response to immune attack, chronic neurodegenerative disease, or acute trauma.
Our work focuses on the mechanisms that induce different forms of reactive astrocytes, and how these reactive cells interact with other cells in the CNS in a positive or negative way. We use high throughput single cell and bulk RNA sequencing, and spatial transcriptomics to investigate the heterogeneity of astrocytes in multiple species. We also take advantage of genetic engineering and modern in vitro modeling to interrogate disease mechanisms and interaction with other CNS cells that change between health and disease.
We apply this knowledge to reactive astrocytes in human disease, and believe that the discovery of astrocytes with different reactive states has important implications for the development of new therapies for CNS injury and diseases. These discoveries have significant implications for neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Amyotrophic Lateral Sclerosis, glaucoma, stroke, and many other neurological conditions.
Ultimately, we aim to provide a more comprehensive understanding of what astrocytes do in disease and how we might ameliorate disease by targeting astrocytes.
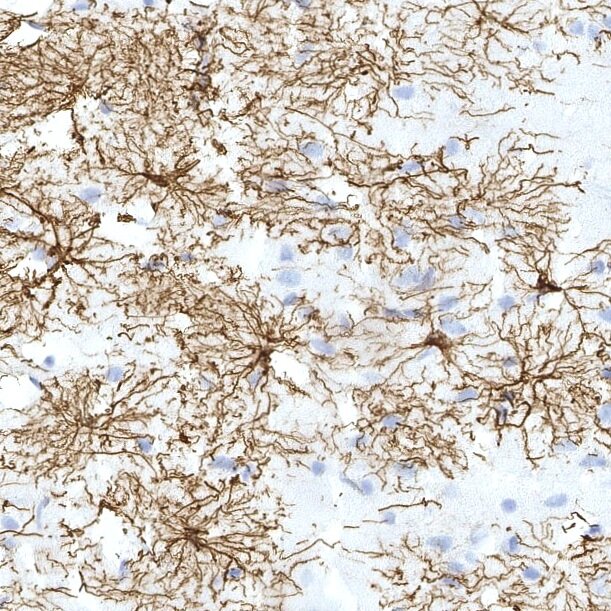
Astrocytes (stained with GFAP, brown) in the human brain. Here the star-shaped morphology of each cell is clearly visible. Image credit: Jessica Sadick.
We are committed to creation and maintenance of an equitable environment in our lab, our Institution, and our University. We oppose racism and hate through allyship, continuous unlearning, learning and listening. We believe excellence in science, respect of people, and rigor of values. Read more about our beliefs, values, and action plan here.

Image credit to Smith Lab @ University of Notre Dame
We are forever grateful to those who fund our work, including the National Institutes of Health (National Eye Institute, National Institute of Neurological Disorders and Stroke, National Institute on Aging), the Cure Alzheimer’s Fund, the Alzheimer’s Association, The Belfer Neurodegeneration Consortium, the Carol and Gene Ludwig Family Foundation, the National Multiple Sclerosis Society, the Collaborative Center for X-Linked Dystonia-Parkinsonism at Mass General Hospital, the Pershing Square Foundation MIND Prize, the Advanced Research Projects Agency for Health (ARPA-H), the Leon Levy Foundation, and NYU Grossman School of Medicine.
We also work closely with the Parekh Center for Interdisciplinary Neurology to bring together physicians and scientists to investigate common mechanisms across neurodegenerative disorders, and the Dark Matter Project at the Center for Synthetic Regulatory Genomics at NYU Langone Health.
A list of our previous funders can be found here.

SOLANGE DE LA VEGA MULET SURP Student 2026
SELECTED PUBLICATIONS
(The most up-to-date list is at Pubmed)
Cooper ML, Selles MC, Cammer M, Redd C, Gildea HK, Sall J, Chiurri KE, Cheung P, Wheeler DG, Saab AS, Liddelow SA#, Chao MV# (2026) Astrocytes connect specific brain regions through plastic networks. Nature PMID: 42020738.
Scientific and media coverage
Science coverage: Astrocyte networks span large swaths of brain. The Transmitter, September 3, 2025
Science coverage: Brain support cells form previously unknown network. NYU Langone Health News Hub, April 22, 2026
Science coverage: Gap junctions link up brain-wide astrocyte networks. AlzForum, April 28, 2026.
Media coverage: Newfound brain network is a ‘secret system’ made of helper cells. Nature, April 22, 2026
Media coverage: Astrocytes build long-range communication networks in the brain. Neuroscience News, April 22, 2026
Hasel P, Cooper ML, Marchildon AE, Rufen-Blanchette U, Kim RD, Ma TC, Groh AMR, Hill EJ, Lewis EM, Januszewski M, Light SEW, Smith CJ, Stratton JA, Sloan SA, Kang UJ, Chao MV, Liddelow SA (2025) Defining the molecular identity and morphology of glia limitans superficialis astrocytes in mouse and human. Cell Rep 44(3):115344. PMID: 39982817.
Scientific and media coverage
Science coverage: Madolyn Bowman Rogers. Border Surprise: glia limitans astrocytes sit on brain surface. AlzForum, November 4, 2023
Science coverage: Lauren Shenkman. Unexpected astrocyte gene flips image of brain’s ‘stalwart sentinels’. The Transmitter, March 28, 2025
Prakash P, Erdjument-Bromage H, O'Dea MR, Munson CN, Labib D, Fossati V, Neubert TA, Liddelow SA (2023) Proteomic profiling of interferon-responsive reactive astrocytes in rodent and human. Glia 72(3):625-642. PMID: 38031883.
Guttenplan KA, Weigel MK*, Prakash P*, Wijewardhane PR, Hasel P, Rufen-Blanchette U, Münch AE, Blum JA, Fine J, Neal MC, Bruce KD, Gitler AD, Chopra G, Liddelow SA#, Barres BA# (2021) Neurotoxic reactive astrocytes induce cell death via saturated lipids. Nature 599:102–107. PMID: 34616039.
Scientific and media coverage
Research highlight: Jamie Talan (2021) Reactive astrocytes and Alzheimer’s disease. J Alz Dis.
Research Highlight: Tom Fagan. ELOVL hurts—enzyme makes lipids that turn astrocytes toxic. AlzForum, October 7, 2021.
Media coverage: Shira Polan. Toxic fatty acids to blame for brain cell death after initial injury. NYU Langone Health October 6, 2021.
Data available online here (proteomic, lipidomic, and metabolomic datasets).
Hasel P, Rose IVL, Sadick JS, Kim RD, Liddelow SA (2021) Neuroinflammatory astrocyte subtypes in the mouse brain. Nat Neurosci 24:1475-1487. PMID: 34413515.
Scientific and media coverage
Research Highlight: High-res spatial transcriptomics offers new views of mouse brain. Alzforum, August, 2022.
Data available online here (bulk RNAseq and nanostring) and here (single cell and spatial data).
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung W-S, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Dawson VL, Dawson TM, Buckwalter MS, Rowitch DH, Stevens B, Barres BA (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541:481-487. PMID: 28099414.
Scientific and media coverage
Research Highlight: Yates D (2017) A toxic reaction. Nat Rev Neurosci 18:130.
Research Highlight: Ridler C (2017) Microglia-induced reactive astrocytes – toxic players in neurological disease? Nat Rev Neurol 13:127.
Media Coverage: Pagan Kennedy. An ancient cure for Alzheimer’s? New York Times July 14, 2017.
Media Coverage: Carolyn Gregoire. Brain cells of ‘Villainous Character’ might explain diseases like Parkinson’s. Huffington Post January 25, 2017.

Astrocytes (green: Aldh1l1-eGFP, red: GFAP) have many processes that extend from their cell bodies and wrap around blood vessels. Nuclei are shown in blue (DAPI). Image Credit: Rachel Kim.
POSTDOCTORAL RESEARCHERS
Candidates with an excellent research record and an excitement for asking tough questions should email Shane with a brief introduction, CV, and the names and contact information of at least two references.
GRADUATE STUDENTS
Graduate students will need to apply to a Graduate Training program at NYU before joining the lab. More information on options for Graduate Training in Neuroscience at NYU can be found here.
We are a part of the Institute for Translational Neuroscience, the Departments of Neuroscience and of Ophthalmology. We are affiliated with the Parekh Center for Interdisciplinary Neurology and the Optimal Aging Institute @ NYU Grossman School of Medicine in New York City.
Want to know more about the lab, or glia in general? Drop us a note - we'd love to hear from you.
liddelowlab@gmail.com